All right, let’s get back to it.
Let’s dive into some pieces of sol-gel chemistry.
What is sol-gel chemistry?
It’s the branch of chemistry that makes aerogels.
Sol-Gel Chemistry means a solution of either a metal or carbon-based precursor with ethers to form a polymer.
This polymer will create a vast network of polymers bonded to each other, creating a gel!
Sol → Solution
Gel → Gel
Here’s the vocabulary for beginners:
Polymers are a chain of many repeating/identical large molecules that make up a material.
Precursors are chemical compounds that participate in a chemical reaction to produce a new compound/molecule.
Precursors are like the ones to start arguing, which ends up in a school fight.
Now, for the actual chemistry part.
The Process! 🔑
To create silica aerogels, the “sol” starts as a liquid solution of colloidal nanoparticles.
A colloid (colloidal) is a particle that is in the range of 1 nanometer to 100 nanometers in diameter/size. But because these small particles are in a liquid (suspension), we call this a colloidal suspension.
A suspension is a mixture where the composition isn't the same in every single place of the mixture (heterogeneous). In this heterogeneous mixture, there is a distributed solid in a liquid. The solid in this case are the particles (colloid).
The liquid in the gel makes sure that the solid network of particles doesn’t collapse, and the solid particles make sure the liquid doesn’t escape.
More information in a new article later.
In most cases, the precursors are used to begin creating hydrophilic silica aerogel.
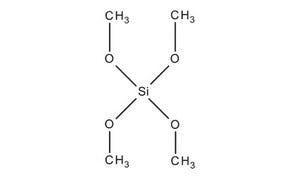
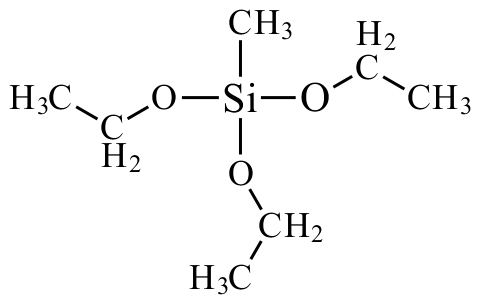
The precursors are either tetramethoxysilane (TMOS) or tetraethoxysilane (TEOS).
The main precursor in creating hydrophobic silica aerogel is commonly known as methyltriethoxysilane (MTES).
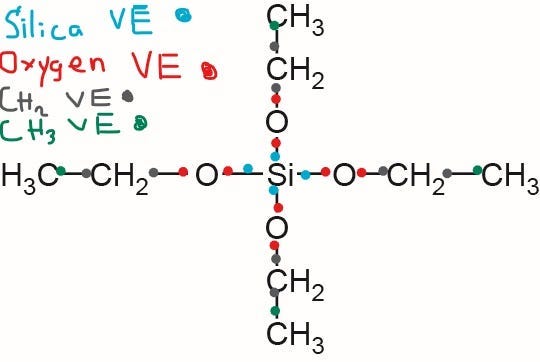
However, the silica atom has a partial positive charge (or lack of negative charge due to loss of negative charge) (δ+) ≈ 0.32 in TEOS.
This means that the silicon atom (Si) tends to lose its Valence Electrons (VE) in its outer shell to the oxygen atoms attached to the silicon atom.
Oxygen has 6 valence electrons, and it takes one electron from silicon and methylene (CH2), which makes 8 valence electrons and fills up its outer shell.
This means that the negative charge is shifting away from the silicon atom in TEOS Precursor.
Bonus:
CH2 has six valence electrons, and it “borrows” two valence electrons with a methyl group (CH3) and Oxygen.
Both the Carbon atom and Hydrogen Atoms have their outer electron shells full.
CH3 has seven valence electrons in its outer shells.
It “borrows” an electron with CH2 to fulfill the carbon and hydrogen outer electron shells.
DONE.
This is how the gel is built for the base.
Next article, we’ll talk about hydrolysis and catalysts can help us in the gel processing of silica aerogels! ✌🏽
With energy,
Carlos ⚛️
Know a friend/colleague that likes aerogels (or bite-sized content)?
Sign up to receive notes about the future of aerogels: